The chronic approach to stopping bladder cancer recurrence
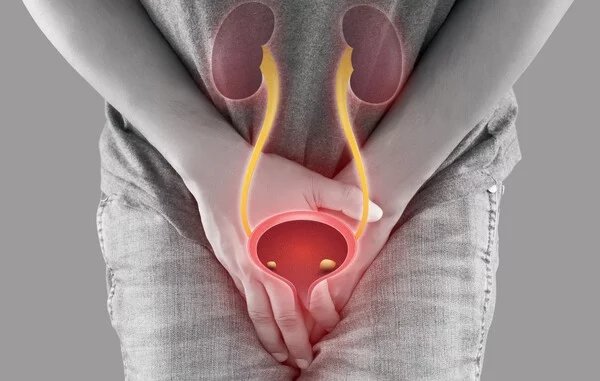
Intravesical therapy delivering anticancer drugs directly into the bladder is a cornerstone treatment for non-muscle-invasive bladder cancer (NMIBC).
While the method may sound unfamiliar, it has been clinically validated for decades and remains central to preventing recurrence and disease progression, News.Az reports, citing Korea Biomed.
Bladder cancer is the ninth most common malignancy worldwide. As of 2022, there were about 610,000 new cases globally, accounting for 3.1 percent of all cancers, representing a significant social and economic burden.
Bladder cancer is classified into non-muscle-invasive (NMIBC) and muscle-invasive (MIBC) types depending on whether the tumor has penetrated the muscle layer of the bladder wall.
RECOMMENDED STORIES
At initial diagnosis, about 75 percent of cases are NMIBC. However, NMIBC is not simply “early-stage” disease. Based on risks of recurrence and progression, it is further stratified into low-, intermediate-, high-, and very high-risk groups.
In high- and very high-risk patients, five-year recurrence rates can reach up to 78 percent, and progression to muscle-invasive disease can be as high as 44 percent—highlighting the need for active, long-term management
Intravesical therapy involves inserting a thin catheter through the urethra into the bladder and instilling anticancer or immunotherapeutic agents directly into the bladder. The drug remains in place for a set period, contacting tumor cells before being expelled through urination. Its primary role is to eliminate residual tumor cells and prevent recurrence after transurethral resection of bladder tumor (TURBT).
After TURBT, a single instillation within 24 hours is typically performed to prevent implantation of residual tumor cells. This is followed by outpatient treatment consisting of induction and maintenance therapy. Induction therapy usually involves weekly instillations for six weeks, while maintenance therapy is essential for high-risk patients.
The rationale for intravesical therapy is straightforward. Because the bladder is directly accessible via the urethra, high concentrations of therapeutic agents can be delivered locally without systemic exposure.
This approach offers three major advantages. First, it enables high local drug concentrations, often exceeding those achievable through systemic therapy. Second, systemic toxicity is significantly reduced. Due to the bladder’s mucosal barrier, blood drug levels remain low, minimizing side effects such as bone marrow suppression, nausea, vomiting, and hair loss. Third, it reduces recurrence and disease progression, supporting bladder preservation and quality of life.
Agents used in intravesical therapy are broadly categorized into immunotherapies and chemotherapies. Among them, Bacillus Calmette-Guérin (BCG) remains the most established standard for high-risk NMIBC following TURBT.
Among chemotherapeutic agents, mitomycin C (MMC) and gemcitabine are widely used. MMC inhibits tumor cell division through DNA crosslinking and is commonly used either immediately after TURBT or as part of induction and maintenance therapy. Gemcitabine is often used in patients who are intolerant to or have failed BCG therapy and shows comparable efficacy to MMC.
While single-agent therapy has limitations—particularly after BCG failure—combination regimens are showing improved outcomes. For example, gemcitabine plus docetaxel achieved a one-year recurrence-free survival rate of 65 percent, while a triple combination of cabazitaxel, gemcitabine, and cisplatin reached 83 percent. In a European multicenter cohort, sequential gemcitabine and cisplatin achieved 73 percent one-year disease-free survival and 96 percent progression-free survival.
Although these combination approaches are not yet widely adopted in Korea and may carry higher complication risks, they are likely to play an increasing role in future treatment strategies.
Most side effects of intravesical therapy are localized and temporary. Common symptoms include burning during urination, urinary frequency, and urgency, which usually resolve within a few days. Mitomycin C may cause chemical cystitis or skin rash, while gemcitabine is generally well tolerated. Treatment should be postponed in patients with active urinary tract infections or bladder injury.
New intravesical therapies are rapidly emerging
Nadofaragene firadenovec, a gene therapy delivering interferon alfa-2b via an adenoviral vector, has received FDA approval for this indication. Tumor-selective viral therapies such as CG0070 are also showing promise. TAR-200, a sustained-release gemcitabine delivery device, achieved a complete response rate of 76.7 percent in patients with BCG-unresponsive carcinoma in situ and has received FDA Breakthrough Therapy designation.
With multiple therapies under development, bladder-preserving treatment strategies are expected to evolve rapidly.
Although NMIBC is often perceived as early-stage cancer, it behaves more like a chronic disease due to its high recurrence and progression risk. Management therefore extends beyond tumor removal to long-term disease control and quality of life. Intravesical therapy remains central to this approach, and treatment should be tailored to individual patient risk and response.
By Leyla Şirinova



























