Abu Dhabi first to deliver ITVISMA gene therapy
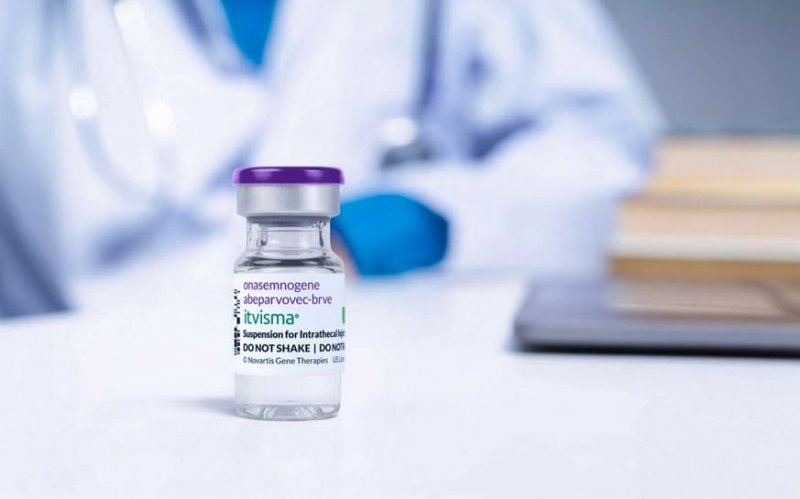
Abu Dhabi has become the first city in the world to administer ITVISMA (onasemnogene abeparvovec), a breakthrough gene therapy for spinal muscular atrophy (SMA). The treatment was successfully delivered at Sheikh Khalifa Medical City under the supervision of the Department of Health – Abu Dhabi.
Developed by Novartis, ITVISMA is a one-time therapy for patients aged 2 and older with a confirmed SMN1 gene mutation. The therapy received accelerated approval in the UAE on November 25, 2025, making the country among the first globally — after the United States — to adopt it, News.Az reports, citing WAM.
Officials highlighted the milestone as a reflection of Abu Dhabi’s commitment to genomics, precision medicine, and access to advanced therapies for rare diseases. Dr. Noura Khamis Al Ghaithi of the Department of Health called it a “significant step in providing cutting-edge care for patients with neuromuscular disorders.”
Bader Al Qubaisi, CEO of SKMC, emphasized the emirate’s integrated healthcare ecosystem, while Novartis’ Mohamed Ezz Eldin said the therapy would have a profound impact on patients and families, strengthening Abu Dhabi’s role as a reference center for advanced neuromuscular care.
As 2025 ends, this achievement adds to a year of global scientific breakthroughs, from gene-editing innovations to faster epidemic response and biodiversity gains, highlighting the power of research, policy coordination, and international collaboration.